Fast prototyping of epidemiological models based on reaction equations. Analyze the ODEs analytically or numerically, or run stochastic simulations on networks/well-mixed systems.
Project description

Fast prototyping of epidemiological models based on reaction equations. Analyze the ODEs analytically or numerically, or run/animate stochastic simulations on networks/well-mixed systems.
repository: https://github.com/benmaier/epipack/
documentation: http://epipack.benmaier.org/
import epipack as epk
from epipack.vis import visualize
import netwulf as nw
network, _, __ = nw.load('cookbook/readme_vis/MHRN.json')
N = len(network['nodes'])
links = [ (l['source'], l['target'], 1.0) for l in network['links'] ]
S, I, R = list("SIR")
model = epk.StochasticEpiModel([S,I,R],N,links)\
.set_link_transmission_processes([ (I, S, 1.0, I, I) ])\
.set_node_transition_processes([ (I, 1.0, R) ])\
.set_random_initial_conditions({ S: N-5, I: 5 })
visualize(model, network, sampling_dt=0.1)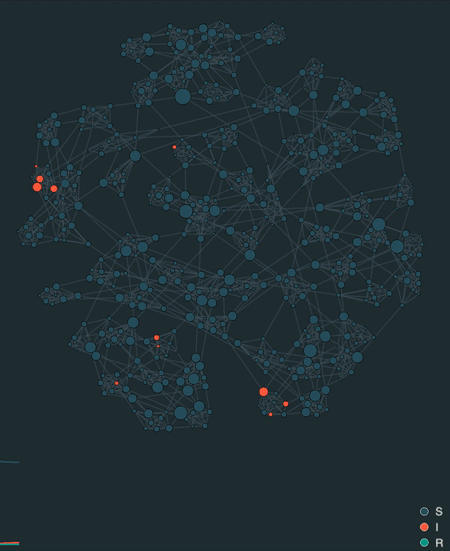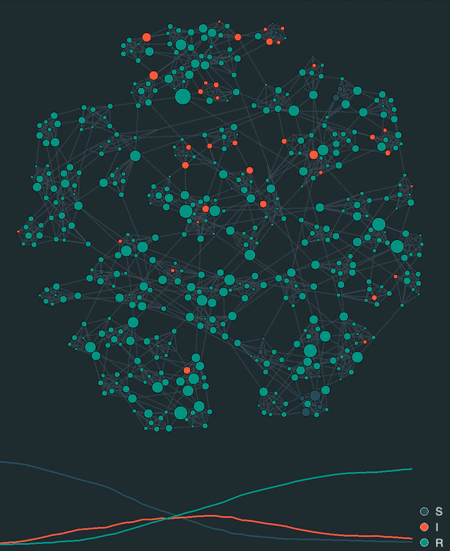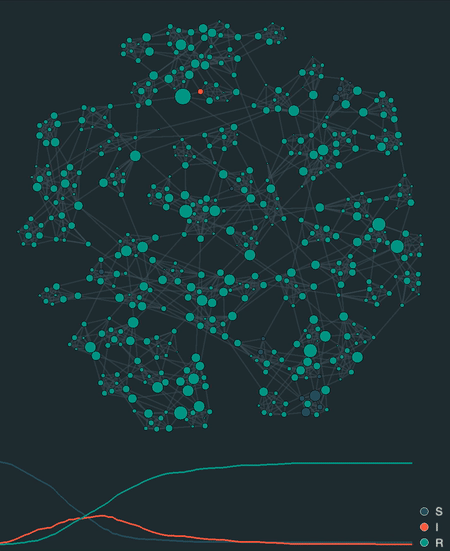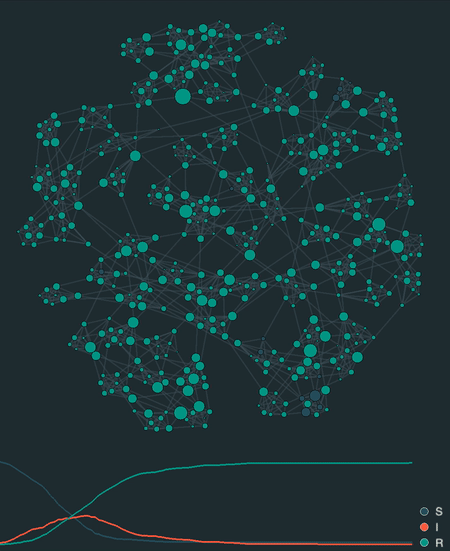
Idea
Simple compartmental models of infectious diseases are useful to investigate effects of certain processes on disease dissemination. Using pen and paper, quickly adding/removing compartments and transition processes is easy, yet the analytical and numerical analysis or stochastic simulations can be tedious to set up and debug—especially when the model changes (even slightly). epipack aims at streamlining this process such that all the analysis steps can be performed in an efficient manner, simply by defining processes based on reaction equations. epipack provides three main base classes to accomodate different problems.
EpiModel: Define a model based on transition, birth, death, fission, fusion, or transmission reactions, integrate the ordinary differential equations (ODEs) of the corresponding well-mixed system numerically or simulate it using Gillespie’s algorithm. Process rates can be numerical functions of time and the system state.
SymbolicEpiModel: Define a model based on transition, birth, death, fission, fusion, or transmission reactions. Obtain the ODEs, fixed points, Jacobian, and the Jacobian’s eigenvalues at fixed points as symbolic expressions. Process rates can be symbolic expressions of time and the system state. Set numerical parameter values and integrate the ODEs numerically or simulate the stochastic systems using Gillespie’s algorithm.
StochasticEpiModel: Define a model based on node transition and link transmission reactions. Add conditional link transmission reactions. Simulate your model on any (un-/)directed, (un-/)weighted static/temporal network, or in a well-mixed system.
Additionally, epipack provides a visualization framework to animate stochastic simulations on networks, lattices, well-mixed systems, or reaction-diffusion systems based on MatrixEpiModel.
Check out the Example section for some demos.
Note that the internal simulation algorithm for network simulations is based on the following paper:
“Efficient sampling of spreading processes on complex networks using a composition and rejection algorithm”, G.St-Onge, J.-G. Young, L. Hébert-Dufresne, and L. J. Dubé, Comput. Phys. Commun. 240, 30-37 (2019), http://arxiv.org/abs/1808.05859.
Install
pip install epipackepipack was developed and tested for
Python 3.6
Python 3.7
Python 3.8
So far, the package’s functionality was tested on Mac OS X and CentOS only.
Dependencies
epipack directly depends on the following packages which will be installed by pip during the installation process
numpy>=1.17
scipy>=1.3
sympy==1.6
pyglet<1.6
matplotlib>=3.0.0
ipython>=7.14.0
ipywidgets>=7.5.1
Please note that fast network simulations are only available if you install
SamplableSet==2.0 (SamplableSet)
manually (pip won’t do it for you).
Documentation
The full documentation is available at epipack.benmaier.org.
Changelog
Changes are logged in a separate file.
License
This project is licensed under the MIT License. Note that this excludes any images/pictures/figures shown here or in the documentation.
Contributing
If you want to contribute to this project, please make sure to read the code of conduct and the contributing guidelines. In case you’re wondering about what to contribute, we’re always collecting ideas of what we want to implement next in the outlook notes.
Examples
Let’s define an SIRS model with infection rate eta, recovery rate rho, and waning immunity rate omega and analyze the system
Pure Numeric Models
Basic Definition (EpiModel)
Define a pure numeric model with EpiModel. Integrate the ODEs or simulate the system stochastically.
from epipack import EpiModel
import matplotlib.pyplot as plt
import numpy as np
S, I, R = list("SIR")
N = 1000
SIRS = EpiModel([S,I,R],N)\
.set_processes([
#### transmission process ####
# S + I (eta=2.5/d)-> I + I
(S, I, 2.5, I, I),
#### transition processes ####
# I (rho=1/d)-> R
# R (omega=1/14d)-> S
(I, 1, R),
(R, 1/14, S),
])\
.set_initial_conditions({S:N-10, I:10})
t = np.linspace(0,40,1000)
result_int = SIRS.integrate(t)
t_sim, result_sim = SIRS.simulate(t[-1])
for C in SIRS.compartments:
plt.plot(t, result_int[C])
plt.plot(t_sim, result_sim[C])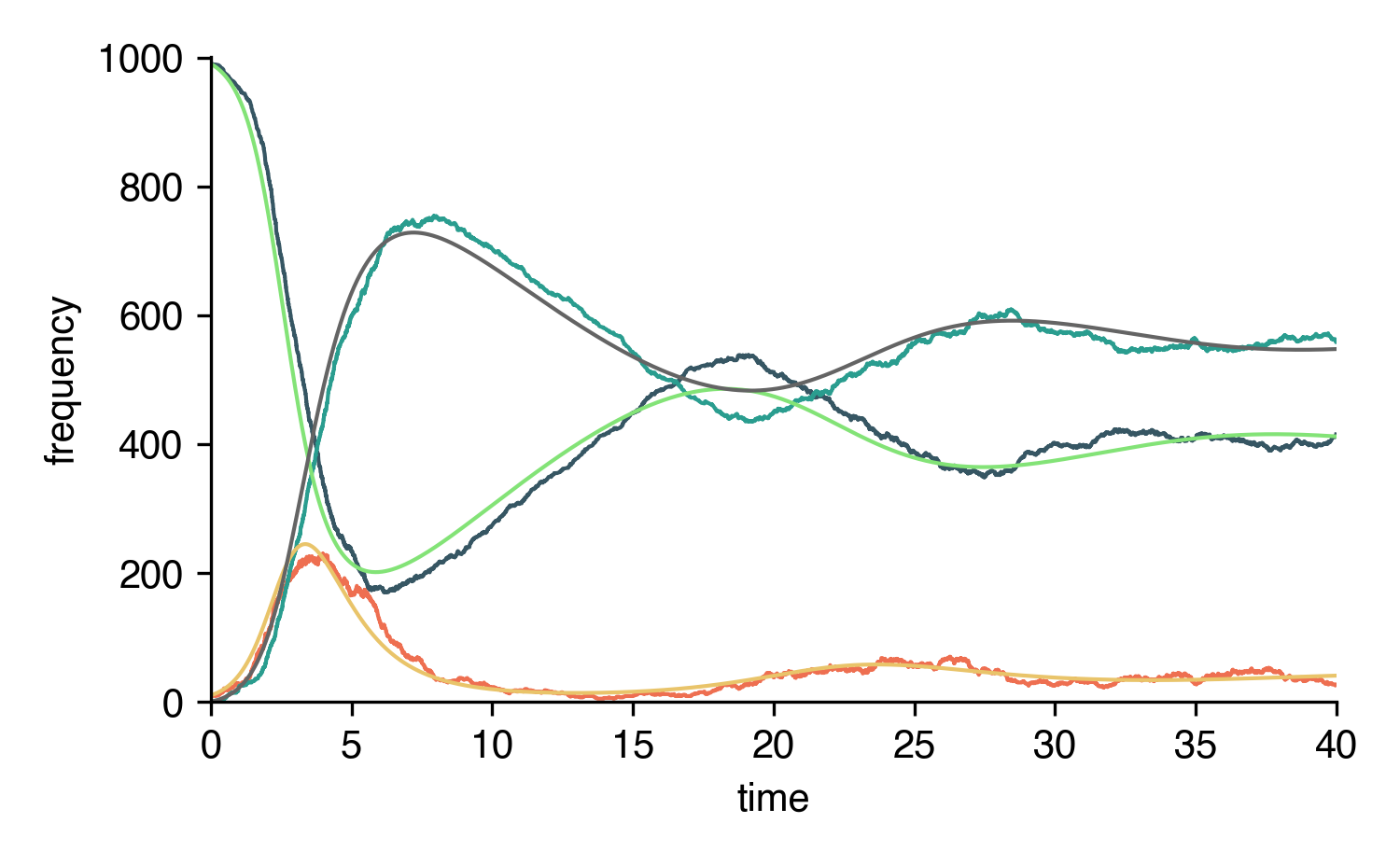
Functional Rates
It’s also straight-forward to define temporally varying (functional) rates.
import numpy as np
from epipack import SISModel
N = 100
recovery_rate = 1.0
def infection_rate(t, y, *args, **kwargs):
return 3 + np.sin(2*np.pi*t/100)
SIS = SISModel(
infection_rate=infection_rate,
recovery_rate=recovery_rate,
initial_population_size=N
)\
.set_initial_conditions({
'S': 90,
'I': 10,
})
t = np.arange(200)
result_int = SIS.integrate(t)
t_sim, result_sim = SIS.simulate(199)
for C in SIS.compartments:
plt.plot(t_sim, result_sim[C])
plt.plot(t, result_int[C])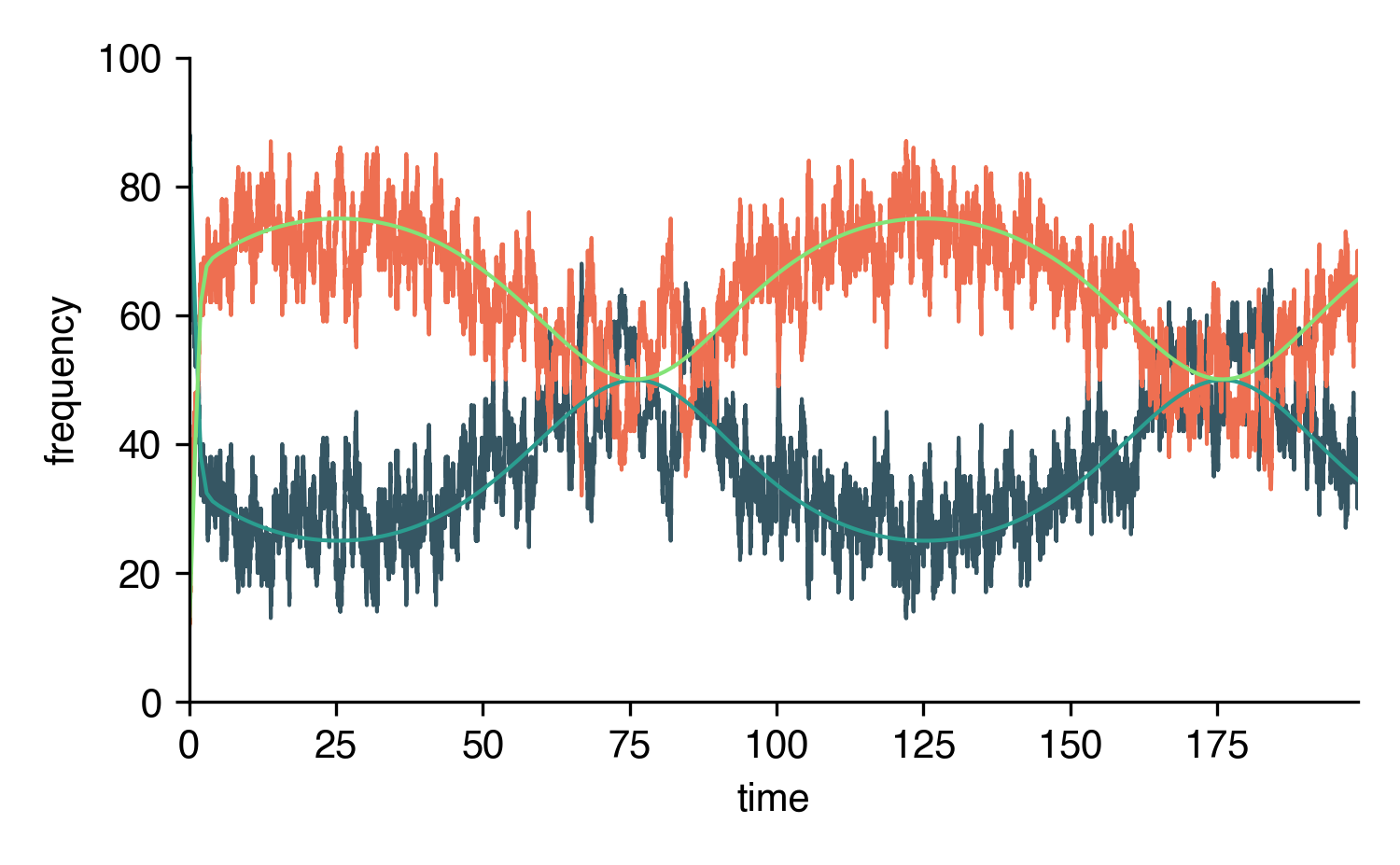
Symbolic Models
Basic Definition
Symbolic models are more powerful because they can do the same as the pure numeric models while also offering the possibility to do analytical evaluations
from epipack import SymbolicEpiModel
import sympy as sy
S, I, R, eta, rho, omega = sy.symbols("S I R eta rho omega")
SIRS = SymbolicEpiModel([S,I,R])\
.set_processes([
(S, I, eta, I, I),
(I, rho, R),
(R, omega, S),
])Analytical Evaluations
Print the ODE system in a Jupyter notebook
>>> SIRS.ODEs_jupyter()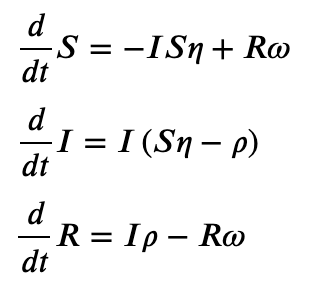
Get the Jacobian
>>> SIRS.jacobian()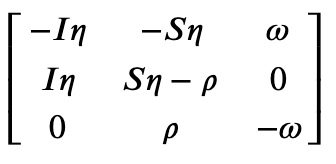
Find the fixed points
>>> SIRS.find_fixed_points()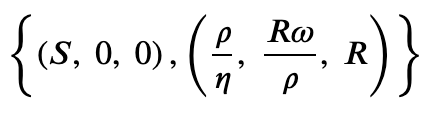
Get the eigenvalues at the disease-free state in order to find the epidemic threshold
>>> SIRS.get_eigenvalues_at_disease_free_state()
{-omega: 1, eta - rho: 1, 0: 1}Numerical Evaluations
Set numerical parameter values and integrate the ODEs numerically
>>> SIRS.set_parameter_values({eta: 2.5, rho: 1.0, omega:1/14})
>>> t = np.linspace(0,40,1000)
>>> result = SIRS.integrate(t)If set up as
>>> N = 10000
>>> SIRS = SymbolicEpiModel([S,I,R],N)the system can simulated directly.
>>> t_sim, result_sim = SIRS.simulate(40)Temporally Varying Rates
Let’s set up some temporally varying rates
from epipack import SymbolicEpiModel
import sympy as sy
S, I, R, eta, rho, omega, t, T = \
sy.symbols("S I R eta rho omega t T")
N = 1000
SIRS = SymbolicEpiModel([S,I,R],N)\
.set_processes([
(S, I, 2+sy.cos(2*sy.pi*t/T), I, I),
(I, rho, R),
(R, omega, S),
])
SIRS.ODEs_jupyter()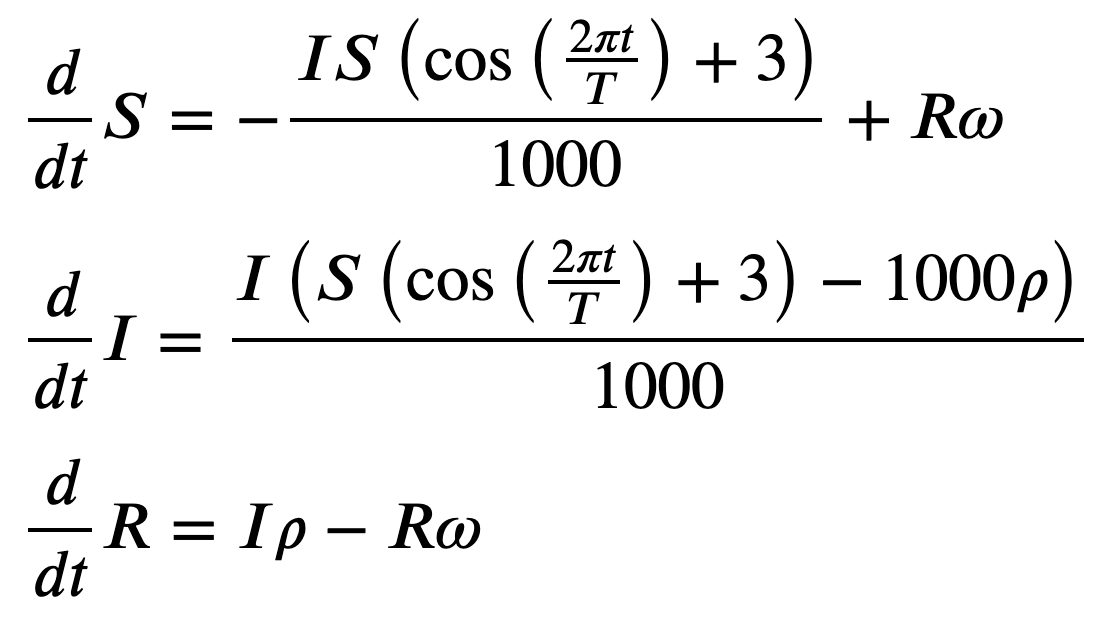
Now we can integrate the ODEs or simulate the system using Gillespie’s SSA for inhomogeneous Poisson processes.
import numpy as np
SIRS.set_parameter_values({
rho : 1,
omega : 1/14,
T : 100,
})
SIRS.set_initial_conditions({S:N-100, I:100})
_t = np.linspace(0,200,1000)
result = SIRS.integrate(_t)
t_sim, result_sim = SIRS.simulate(max(_t))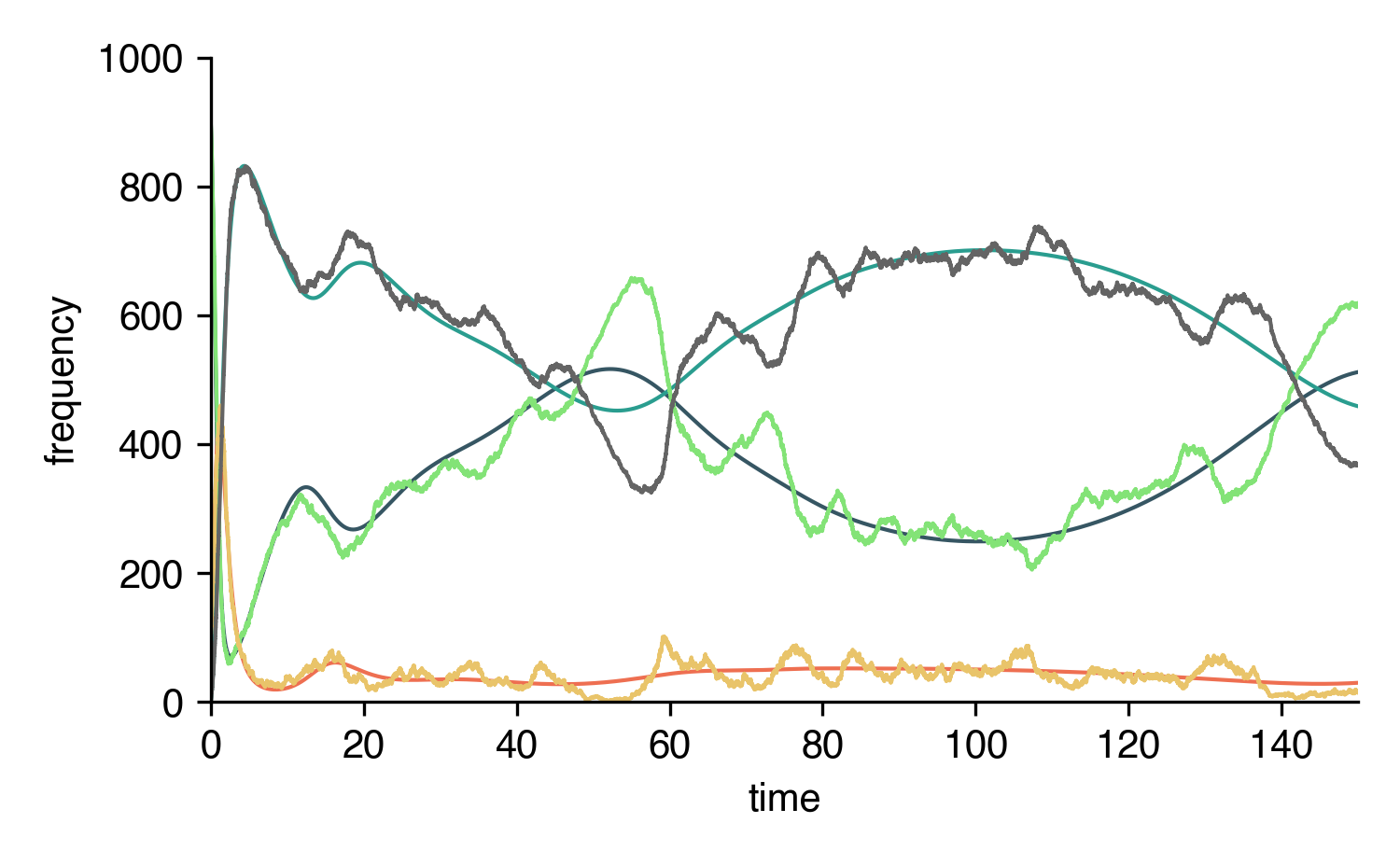
Interactive Analyses
epipack offers a classs called InteractiveIntegrator that allows an interactive exploration of a system in a Jupyter notebook.
Make sure to first run
%matplotlib widgetin a cell.
from epipack import SymbolicEpiModel
from epipack.interactive import InteractiveIntegrator, Range, LogRange
import sympy
S, I, R, R0, tau, omega = sympy.symbols("S I R R_0 tau omega")
I0 = 0.01
model = SymbolicEpiModel([S,I,R])\
.set_processes([
(S, I, R0/tau, I, I),
(I, 1/tau, R),
(R, omega, S),
])\
.set_initial_conditions({S:1-I0, I:I0})
# define a log slider, a linear slider and a constant value
parameters = {
R0: LogRange(min=0.1,max=10,step_count=1000),
tau: Range(min=0.1,max=10,value=8.0),
omega: 1/14
}
t = np.logspace(-3,2,1000)
InteractiveIntegrator(model, parameters, t, figsize=(4,4))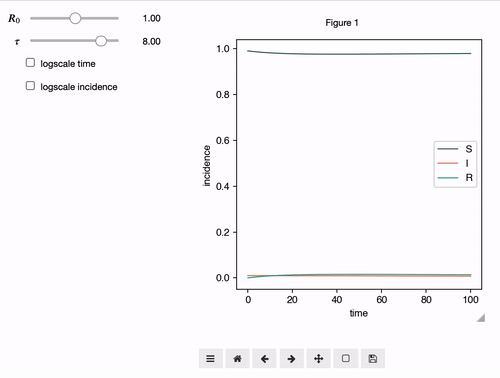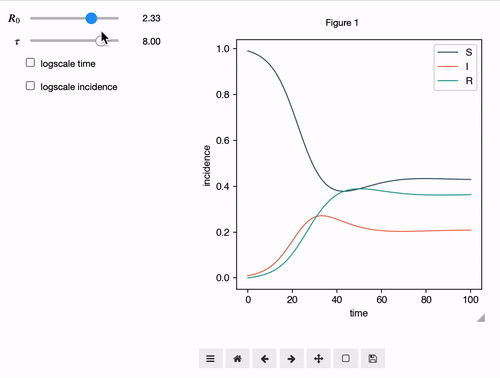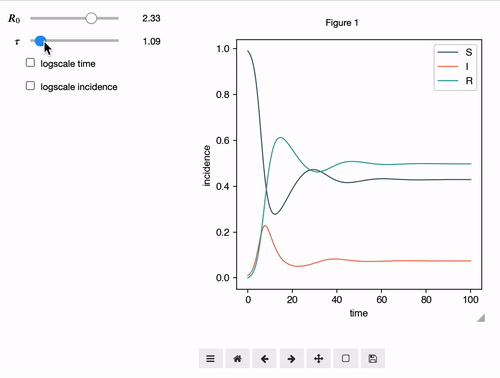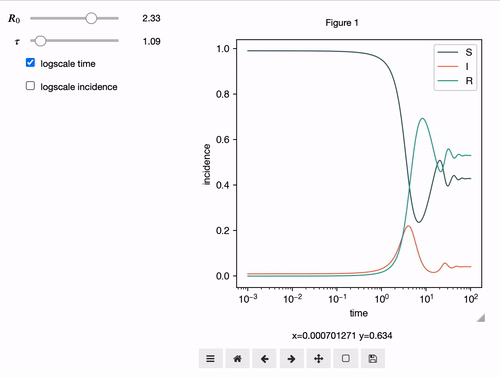
Pure Stochastic Models
On a Network
Let’s simulate an SIRS system on a random graph (using the parameter definitions above).
from epipack import StochasticEpiModel
import networkx as nx
k0 = 50
R0 = 2.5
rho = 1
eta = R0 * rho / k0
omega = 1/14
N = int(1e4)
edges = [ (e[0], e[1], 1.0) for e in \
nx.fast_gnp_random_graph(N,k0/(N-1)).edges() ]
SIRS = StochasticEpiModel(
compartments=list('SIR'),
N=N,
edge_weight_tuples=edges
)\
.set_link_transmission_processes([
('I', 'S', eta, 'I', 'I'),
])\
.set_node_transition_processes([
('I', rho, 'R'),
('R', omega, 'S'),
])\
.set_random_initial_conditions({
'S': N-100,
'I': 100
})
t_s, result_s = SIRS.simulate(40)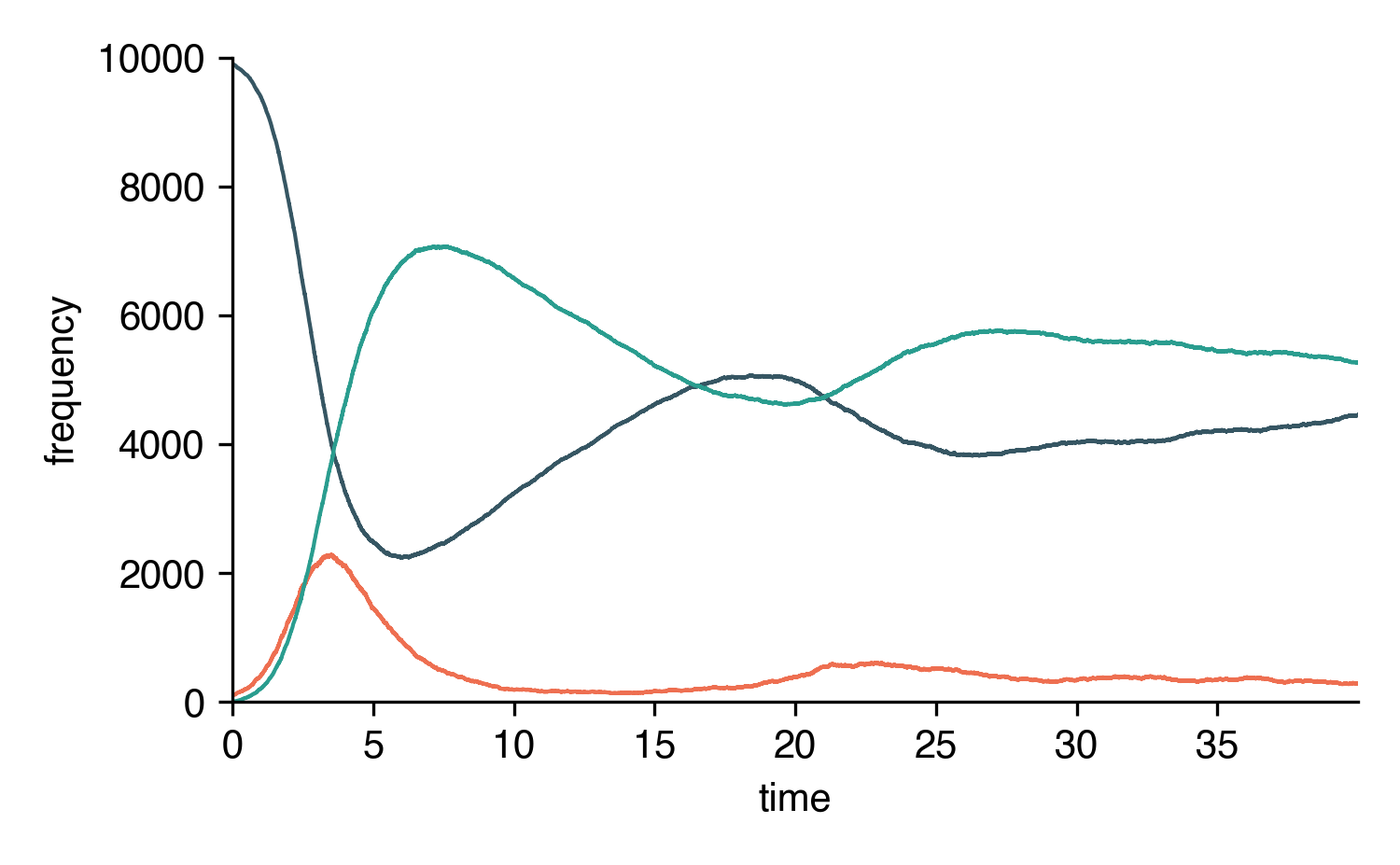
Visualize
Likewise, it’s straight-forward to visualize this system
>>> from epipack.vis import visualize
>>> from epipack.networks import get_random_layout
>>> layouted_network = get_random_layout(N, edges)
>>> visualize(SIRS, layouted_network, sampling_dt=0.1, config={'draw_links': False})
On a Lattice
A lattice is nothing but a network, we can use get_grid_layout and get_2D_lattice_links to set up a visualization.
from epipack.vis import visualize
from epipack import (
StochasticSIRModel,
get_2D_lattice_links,
get_grid_layout
)
# define links and network layout
N_side = 100
N = N_side**2
links = get_2D_lattice_links(N_side, periodic=True, diagonal_links=True)
lattice = get_grid_layout(N)
# define model
R0 = 3; recovery_rate = 1/8
model = StochasticSIRModel(N,R0,recovery_rate,
edge_weight_tuples=links)
model.set_random_initial_conditions({'I':20,'S':N-20})
sampling_dt = 1
visualize(model,lattice,sampling_dt,
config={
'draw_nodes_as_rectangles':True,
'draw_links':False,
}
)
Reaction-Diffusion Models
Since reaction-diffusion systems in discrete space can be interpreted as being based on reaction equations, we can set those up using epipack’s framework.
Checkout the docs on Reaction-Diffusion Systems.
Every node in a network is associated with a compartment and we’re using MatrixEpiModel because it’s faster than EpiModel.
from epipack import MatrixEpiModel
N = 100
base_compartments = list("SIR")
compartments = [ (node, C) for node in range(N) for C in base_compartments ]
model = MatrixEpiModel(compartments)Now, we define both epidemiological and movement processes on a hypothetical list links.
infection_rate = 2
recovery_rate = 1
mobility_rate = 0.1
quadratic_processes = []
linear_processes = []
for node in range(N):
quadratic_processes.append(
( (node, "S"), (node, "I"), infection_rate, (node, "I"), (node, "I") ),
)
linear_processes.append(
( (node, "I"), recovery_rate, (node, "R") )
)
for u, v, w in links:
for C in base_compartments:
linear_processes.extend([
( (u, C), w*mobility_rate, (v, C) ),
( (v, C), w*mobility_rate, (u, C) ),
])
Dev notes
Fork this repository, clone it, and install it in dev mode.
git clone git@github.com:YOURUSERNAME/epipack.git
makeIf you want to upload to PyPI, first convert the new README.md to README.rst
make readmeIt will give you warnings about bad .rst-syntax. Fix those errors in README.rst. Then wrap the whole thing
make pypiIt will probably give you more warnings about .rst-syntax. Fix those until the warnings disappear. Then do
make uploadProject details
Download files
Download the file for your platform. If you're not sure which to choose, learn more about installing packages.
Source Distribution
File details
Details for the file epipack-0.1.5.tar.gz.
File metadata
- Download URL: epipack-0.1.5.tar.gz
- Upload date:
- Size: 93.4 kB
- Tags: Source
- Uploaded using Trusted Publishing? No
- Uploaded via: twine/3.2.0 pkginfo/1.6.1 requests/2.25.0 setuptools/54.2.0 requests-toolbelt/0.9.1 tqdm/4.54.1 CPython/3.8.9
File hashes
| Algorithm | Hash digest | |
|---|---|---|
| SHA256 |
398b8cd08121eb4955afa871723847ab28de57d6d9e98d496fa7ad903c734d52
|
|
| MD5 |
1de0468d88232f17365bf91f7d039131
|
|
| BLAKE2b-256 |
4ffe9763fdaa2f5c14d217a2fa9be0d4e506cf7ef1f9fe9c401d209d0fbd2a66
|












