Retrosynthesis, visualization and machine learning tools
Project description

- RetroChem -
📜 Package information
RetroChem, is a one-step retrosynthesis engine pip installable python package, which is based on SMILES inputs. It includes tools for model training, template preprocessing, and visualization of reaction predictions. Developped as part of a project for the practical Programming in Chemistry at EPFL (2025).
🪄 Features
- Simple to use retrosynthesis engine.
- Draw molecules or use molecule name (eg. paracetamol).
- Gives clean and chemically correct molecules.
- Works well for known molecules (eg. GHB, EDTA and etc).
- Predicts possible reactants and reaction steps.
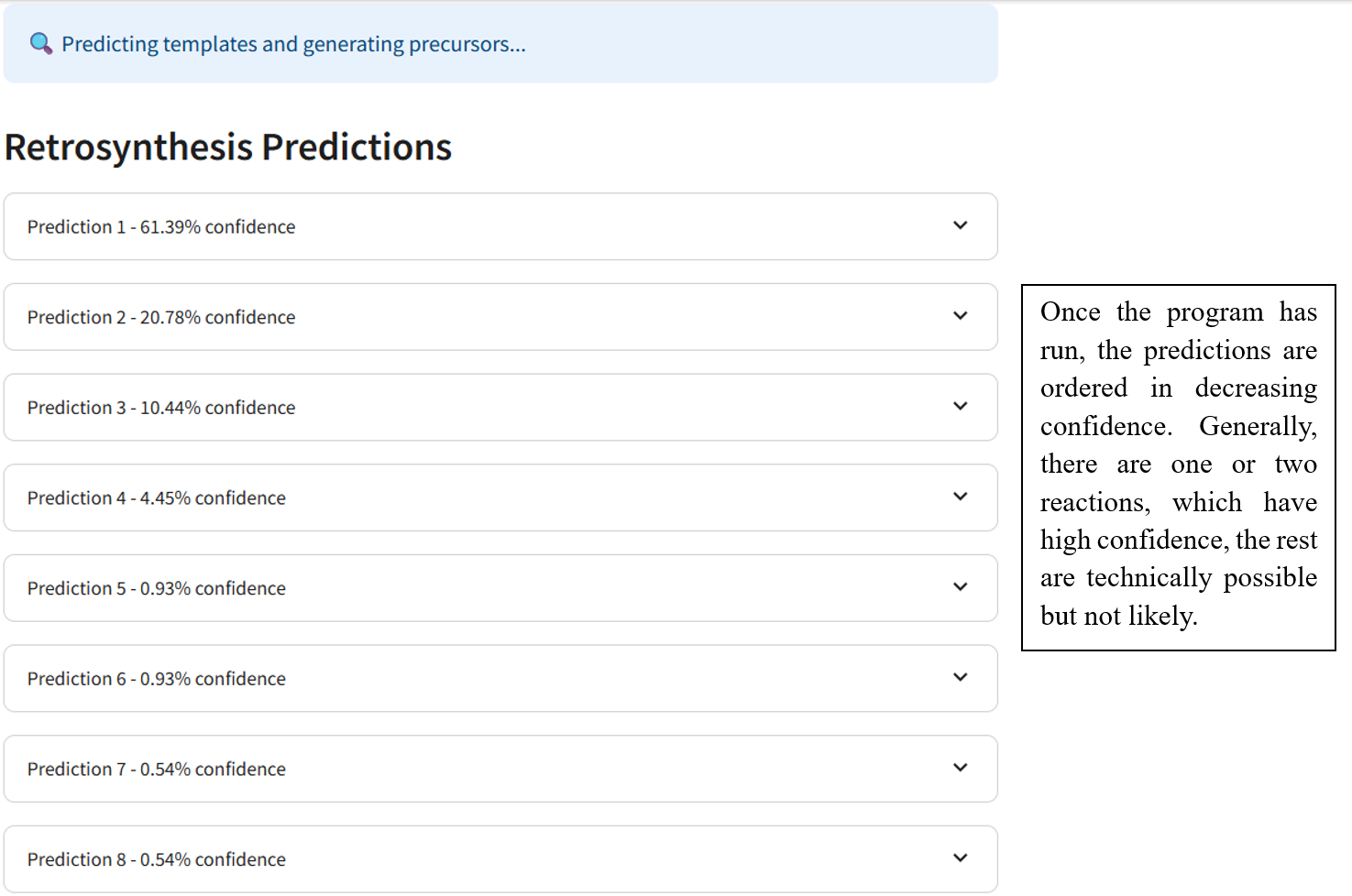
- Gives the predictions in order of confidence
👥 Contributors
- Giulio Matteo Garotti, second year chemical engineer at EPFL
- Florian Follet, second year chemist at EPFL
- Noah Paganzzi, second year chemical engineer at EPFL
- Jacques Maurice Grandjean, second year chemical engineer at EPFL
View contributors : Contributors on GitHub
View commit activity: Commit activity on GitHub
View code frequency : Code frequency on GitHub
🔍 Retrosynthesis, what is it?
Organic retrosynthesis is a problem-solving technique used in organic chemistry to design a synthetic route for a target molecule by breaking it down into simpler precursor structures. This process, known as retrosynthetic analysis, helps chemists plan the step-by-step synthesis of complex organic compounds by working backward from the desired product.
The approach involves two main stages:
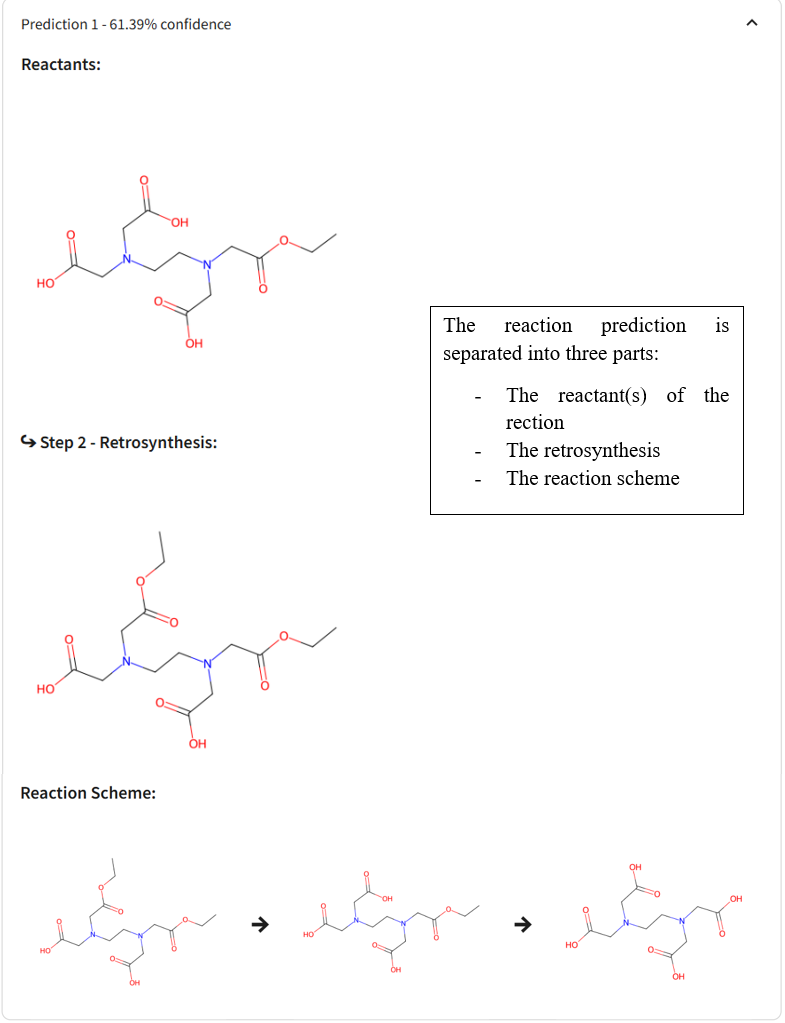
Disconnection: The target molecule is “disconnected” at strategic bonds to identify simpler molecules that could be used as starting materials. These disconnections are guided by known chemical reactions and functional group transformations. Each disconnection leads to one or more synthons, which are idealised fragments representing the functional components of the molecule. For example, a carbon–carbon bond may be disconnected to yield a nucleophile and an electrophile.
Reagent Identification: Once the synthons are defined, they are translated into real, purchasable or synthesizable compounds known as synthetic equivalents. These are then used to build the molecule in the forward direction. This step requires the selection of appropriate reagents, protecting groups, and reaction conditions to ensure a feasible and efficient synthesis.
A retrosynthetic tree is often constructed to visualise all possible synthetic pathways, with the target molecule at the top and potential starting materials branching below. Chemists use both strategic bonds and known reaction patterns (like aldol condensations, Grignard additions, or Diels-Alder reactions) to inform these choices.
- Retrosynthesis is widely applied in:
- Pharmaceutical chemistry, for the development of active pharmaceutical ingredients (APIs).
- Natural product synthesis, for recreating complex bioactive compounds from simpler building blocks.
- Material science, for designing functional molecules with specific properties.
- Green chemistry, to plan more sustainable and efficient synthetic routes.
The logic of retrosynthesis also lends itself well to computational chemistry. Algorithms and machine learning tools are increasingly used to automate retrosynthetic analysis, generating synthetic routes based on reaction databases and predictive models.
The theoretical underpinning of retrosynthesis often involves mapping the retrosynthetic steps to known reaction mechanisms and using heuristics based on reactivity and selectivity. The goal is to find the shortest, most cost-effective, and most reliable pathway to the target compound.
Let us now walk through how to apply retrosynthetic analysis to a real molecule using this framework!
🕹️ How to install it
PYPI page of the RetroChem package:
Firstly it is advised to create a CONDA environment:
#Open bash or terminal
#Name the environment as you wish
conda create -n env.name
#Activate your environment
conda activate env.name
#Alternative way of activating it
source activate env.name
Clone this repository and install the python package locally:
#Clone the repository
git clone https://github.com/Flo-fllt/RetroChem.git
#Naviguate to the RetroChem folder
cd RetroChem
#Install the package locally in editable mode, make sure to activate your environment before doing so
pip install retrochem
#This will install the retrosynth package on your machine. You can now run the program with:
retrochem
The streamlit web page for RetroChem will be opened on your default browser.
💡 Requirements
python>=3.10
streamlit
pandas
numpy
joblib
rdkit
scikit-learn
matplotlib
pillow
streamlit-ketcher
💻 Guide
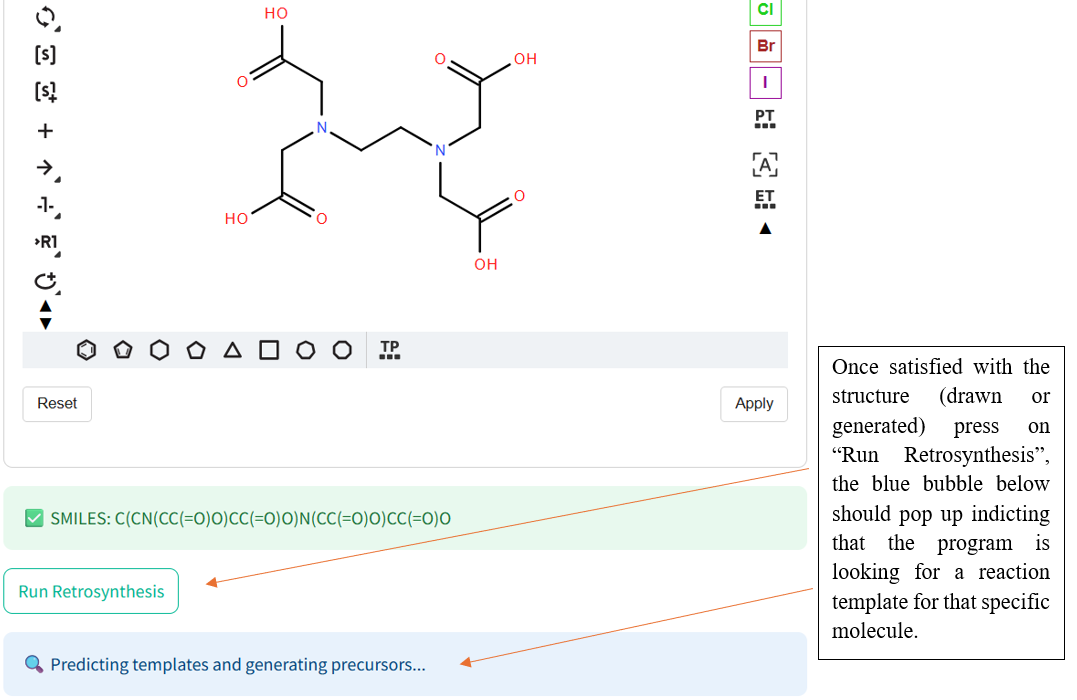
Once the RetroChem web page is open the retrosynthesis engine is ready to run!
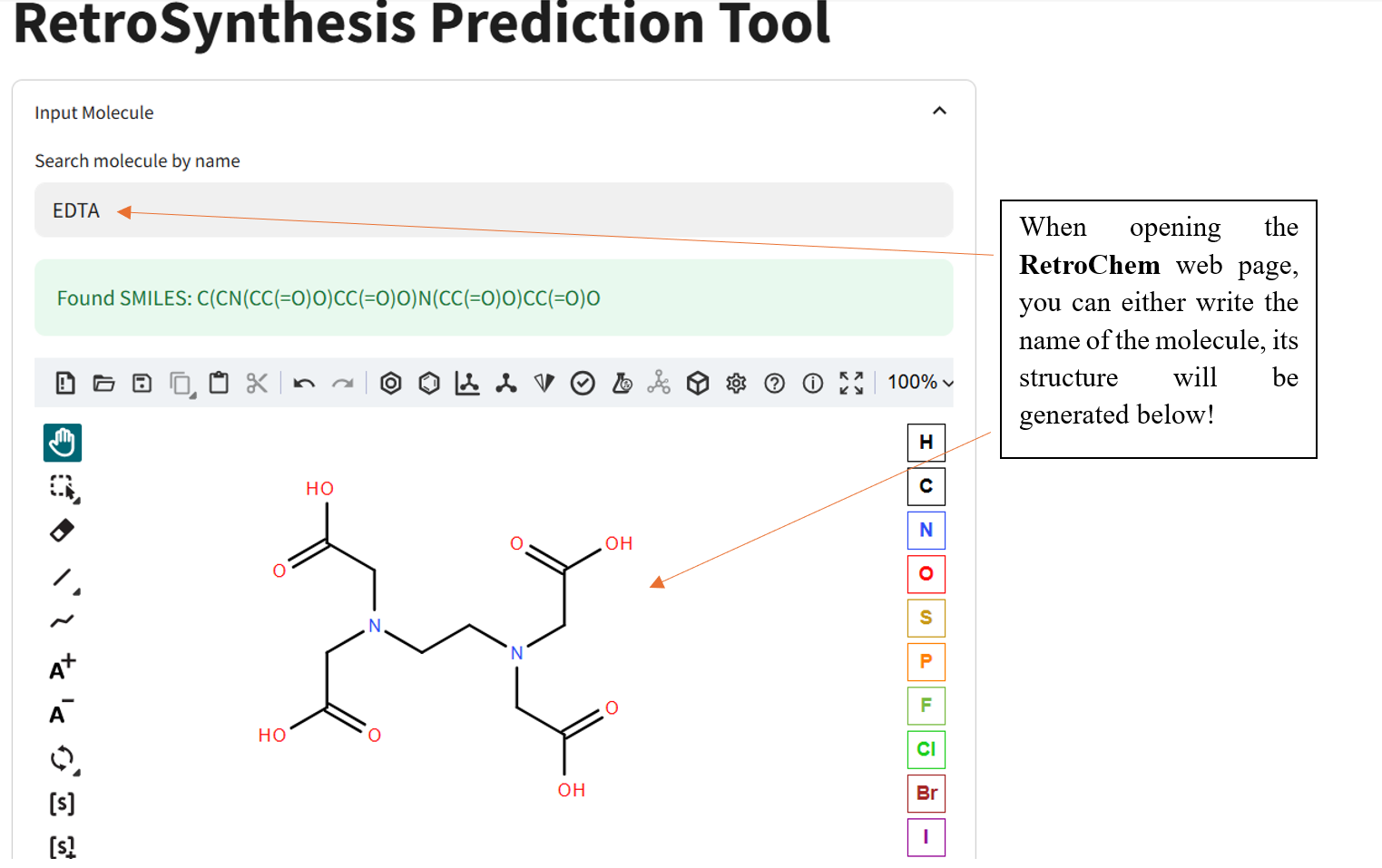
If the program has a difficult time finding reactnts, try another molecule and especially known molecules.
🛟 Need help?
🤖 How to train our model
📚 Want more information?
Here is the link to the report of the project, logging detailed information regarding the functions and data used to efficiently run this program.
Project details
Release history Release notifications | RSS feed
Download files
Download the file for your platform. If you're not sure which to choose, learn more about installing packages.
Source Distribution
Built Distribution
Filter files by name, interpreter, ABI, and platform.
If you're not sure about the file name format, learn more about wheel file names.
Copy a direct link to the current filters
File details
Details for the file retrochem-0.1.9.tar.gz.
File metadata
- Download URL: retrochem-0.1.9.tar.gz
- Upload date:
- Size: 46.5 MB
- Tags: Source
- Uploaded using Trusted Publishing? No
- Uploaded via: twine/6.1.0 CPython/3.10.0
File hashes
| Algorithm | Hash digest | |
|---|---|---|
| SHA256 |
69a4ca06feac168386d809123b7416994b1b2bf68a8dd268599895e28785832f
|
|
| MD5 |
7c63bdca070cb9e8591372789c5f4f53
|
|
| BLAKE2b-256 |
97c92137f5f9d5ad1cf949b9b4691c5834bccee6e27cb178faec65d43b6e09b6
|
File details
Details for the file retrochem-0.1.9-py3-none-any.whl.
File metadata
- Download URL: retrochem-0.1.9-py3-none-any.whl
- Upload date:
- Size: 44.8 MB
- Tags: Python 3
- Uploaded using Trusted Publishing? No
- Uploaded via: twine/6.1.0 CPython/3.10.0
File hashes
| Algorithm | Hash digest | |
|---|---|---|
| SHA256 |
267c17cab5827d3f904dcd18a70c9cee31b7b6261a9c920ea0d5d5e85a5ed542
|
|
| MD5 |
4219259502ef8c5396b3c93d9613f61a
|
|
| BLAKE2b-256 |
0fae2df14c390f0bdfe2b22ef32c308dbf55d95b54b5488a699786f603fe0897
|





























