A multi-sample variant calling pipeline
Project description




This is the variant_calling pipeline from the Sequana projet
- Overview:
Variant calling from FASTQ files
- Input:
FASTQ files from Illumina Sequencing instrument
- Output:
VCF and HTML files
- Status:
production
- Citation:
Cokelaer et al, (2017), ‘Sequana’: a Set of Snakemake NGS pipelines, Journal of Open Source Software, 2(16), 352, JOSS DOI https://doi:10.21105/joss.00352
Installation
You can install sequana_variant_calling pipeline using:
pip install sequana_variant_calling --upgrade
I would recommend to setup a sequana_variant_calling conda environment executing:
conda env create -f environment.yml
where the environment.yml can be found in the https://github.com/sequana/variant_calling repository.
Later, you can activate the environment as follows:
conda activate sequana_variant_calling
Note, however, that the recommended method is to use singularity/apptainer as explained here below.
Usage
sequana_variant_calling --input-directory DATAPATH --reference-file measles.fa
This creates a directory variant_calling. You just need to move into the directory and execute the script:
cd variant_calling sh variant_calling.sh
This launch a snakemake pipeline. If you are familiar with snakemake, you can retrieve the pipeline itself and its configuration files and then execute the pipeline yourself with specific parameters:
snakemake -s variant_calling.rules -c config.yaml --cores 4 --stats stats.txt
you can also edit the profile file in .sequana/profile/config.ya,l
Or use sequanix interface.
Usage with singularity::
With singularity, initiate the working directory as follows:
sequana_variant_calling --use-singularity --singularity-prefix ~/.sequana/apptainers
Images are downloaded in a global direcory (here .sequana/apptainers) so that you can reuse them later.
and then as before:
cd variant_calling sh variant_calling.sh
if you decide to use snakemake manually, do not forget to add singularity options:
snakemake -s variant_calling.rules -c config.yaml --cores 4 --stats stats.txt --use-singularity --singularity-prefix ~/.sequana/apptainers --singularity-args "-B /home:/home"
Requirements
If you rely on singularity/apptainer, no extra dependencies are required (expect python and https://damona.readthedocs.io). If you cannot use apptainer, you will need to install some software:
bwa
freebayes
picard (picard-tools)
sambamba
minimap2
samtools
snpEff you will need 5.0 or 5.1d (note the d); 5.1 does not work.
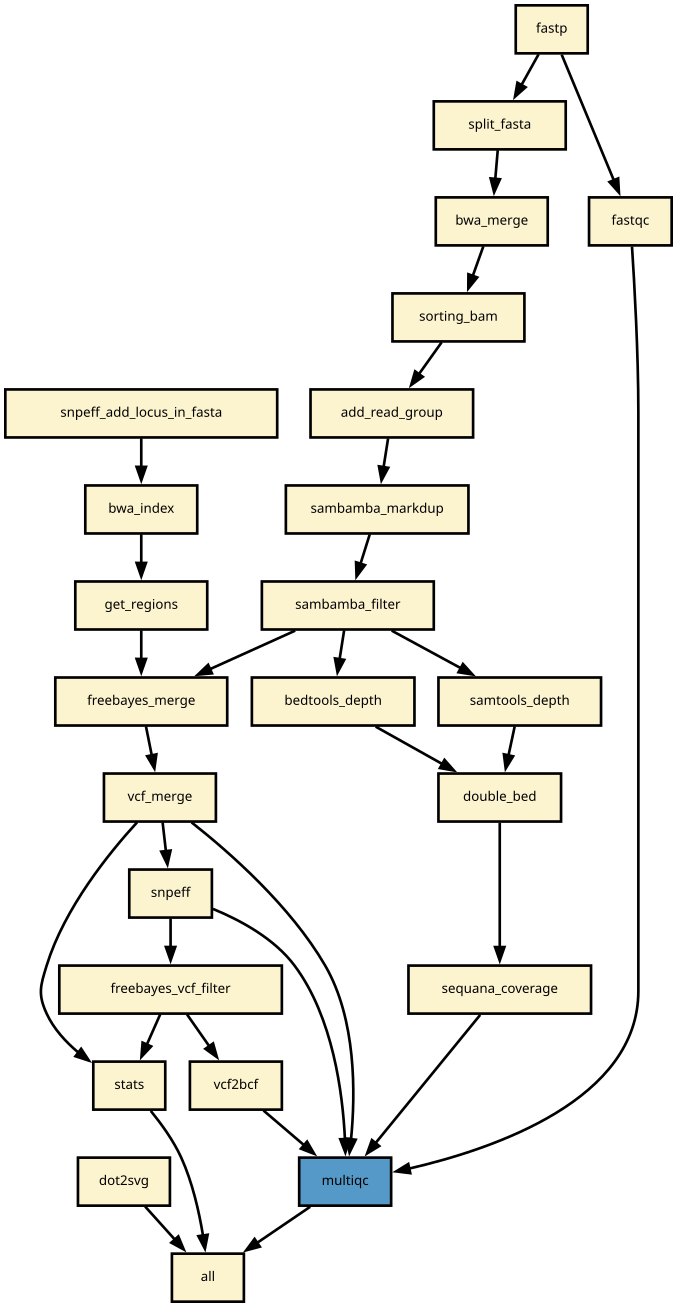
Details
Snakemake variant calling pipeline is based on tutorial written by Erik Garrison. Input reads (paired or single) are mapped using bwa and sorted with sambamba-sort. PCR duplicates are marked with sambamba-markdup. Freebayes is used to detect SNPs and short INDELs. The INDEL realignment and base quality recalibration are not necessary with Freebayes. For more information, please refer to a post by Brad Chapman on minimal BAM preprocessing methods.
The pipeline provides an analysis of the mapping coverage using sequana coverage. It detects and characterises automatically low and high genome coverage regions.
Detected variants are annotated with SnpEff if a GenBank file is provided. The pipeline does the database building automatically. Although most of the species should be handled automatically, some special cases such as particular codon table will required edition of the snpeff configuration file.
Finally, joint calling is also available and can be switch on if desired.
Tutorial
Let us download an ecoli reference genome and the data set used to create the assembly. All tools used here below can be installed with damona (or your favorite environment manager):
pip install damona damona create TEST damona activate TEST damona install pigz damona install sratoolkit # for fasterq-dump damona install datasets
Then, download the data:
fasterq-dump SRR13921546 pigz SRR*fastq
and the reference genome with its annnotation:
datasets download genome accession GCF_000005845.2 --include gff3,rna,cds,protein,genome,seq-report,gbff unzip ncbi_dataset.zip ln -s ncbi_dataset/data/GCF_000005845.2/GCF_000005845.2_ASM584v2_genomic.fna ecoli.fa ln -s ncbi_dataset/data/GCF_000005845.2/genomic.gff ecoli.gff
Initiate the pipeline:
sequana_variant_calling --input-directory . --reference-file ecoli.fa --aligner-choice bwa_split \
--do-coverage --annotation-file ecoli.gff \
--use-apptainer --apptainer-prefix ~/.sequana/apptainers \
--input-readtag "_[12]."
Explication:
we use apptainer/singularity
we use the reference genome ecoli.fa (–reference-file) and its annotation for SNPeff (–annotation-file)
we use the sequana_coverage tool (True by default) to get coverage plots.
we use –input-directory to indicatre where to find the input files
This data set is paired. In NGS, it is common to have _R1_ and _R2_ tags to differentiate the 2 files. Here the tags are _1 and _2. In sequana we define the a wildcard for the read tag. So here we tell the software that thex expected tags follow this pattern: “_[12].” and everything is then automatic.
Then follow the instructions (prepare and execute the pipeline).
You should end up with a summary.hml report.
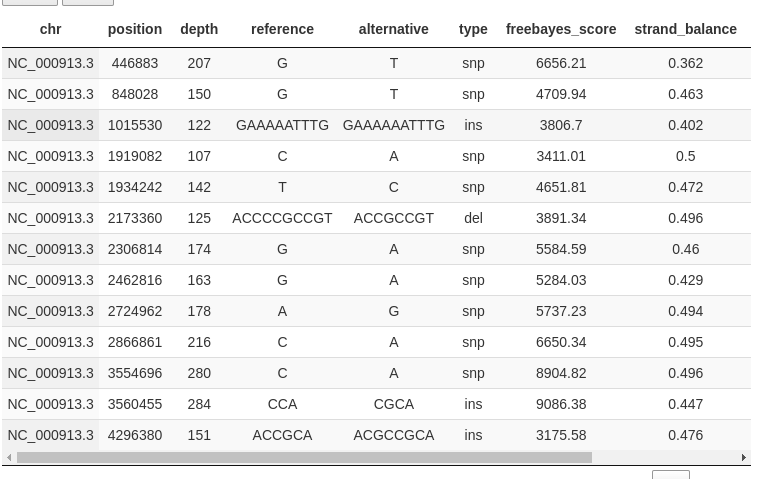
You can browse the different samples (only one in this example) and get a table with variant calls:
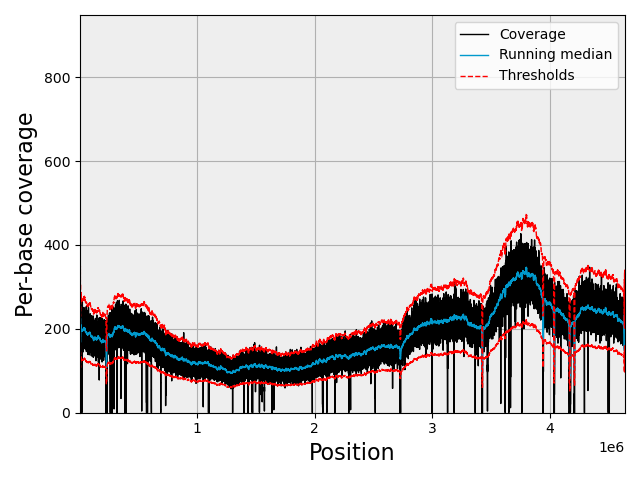
If you set the coverage one, (not recommended for eukaryotes), you should see this kind of plots:
Changelog
Version |
Description |
|---|---|
1.5.0 |
|
1.4.0 |
|
1.3.0 |
|
1.2.0 |
|
1.1.2 |
|
1.1.1 |
|
1.1.0 |
|
1.0.2 |
|
1.0.1 |
|
1.0.0 |
|
0.12.0 |
|
0.11.0 |
|
0.10.0 |
|
0.9.10 |
|
0.9.5 |
|
0.9.4 |
|
0.9.3 |
|
0.9.2 |
|
0.9.1 |
|
0.9.0 |
First release |
Contribute & Code of Conduct
To contribute to this project, please take a look at the Contributing Guidelines first. Please note that this project is released with a Code of Conduct. By contributing to this project, you agree to abide by its terms.
Project details
Release history Release notifications | RSS feed
Download files
Download the file for your platform. If you're not sure which to choose, learn more about installing packages.
Source Distribution
Built Distribution
Filter files by name, interpreter, ABI, and platform.
If you're not sure about the file name format, learn more about wheel file names.
Copy a direct link to the current filters
File details
Details for the file sequana_variant_calling-1.5.0.tar.gz.
File metadata
- Download URL: sequana_variant_calling-1.5.0.tar.gz
- Upload date:
- Size: 122.5 kB
- Tags: Source
- Uploaded using Trusted Publishing? No
- Uploaded via: twine/6.1.0 CPython/3.13.12
File hashes
| Algorithm | Hash digest | |
|---|---|---|
| SHA256 |
ee9294882ef0a0070ad7fc648b05b56999df5cdbd23a45bbde5af8b4f0bf5081
|
|
| MD5 |
65494df62fbf3a89b02d8c4a91ac1663
|
|
| BLAKE2b-256 |
dac45f0c2d627a93e2edf2257facce36a022a07b78a8e26f98030b95b5fa1c07
|
File details
Details for the file sequana_variant_calling-1.5.0-py3-none-any.whl.
File metadata
- Download URL: sequana_variant_calling-1.5.0-py3-none-any.whl
- Upload date:
- Size: 119.9 kB
- Tags: Python 3
- Uploaded using Trusted Publishing? No
- Uploaded via: twine/6.1.0 CPython/3.13.12
File hashes
| Algorithm | Hash digest | |
|---|---|---|
| SHA256 |
145b275b82aae31f2b8aead83aad4ba7832365dae5e976fbba08ccd9b5bf39b5
|
|
| MD5 |
c1e1fbe0e97c2843d034d02a3907b4a5
|
|
| BLAKE2b-256 |
c7e4ef540a5040d0aa2f39602dbce61724972915557dd3c7d3f2c2e4241ea431
|











